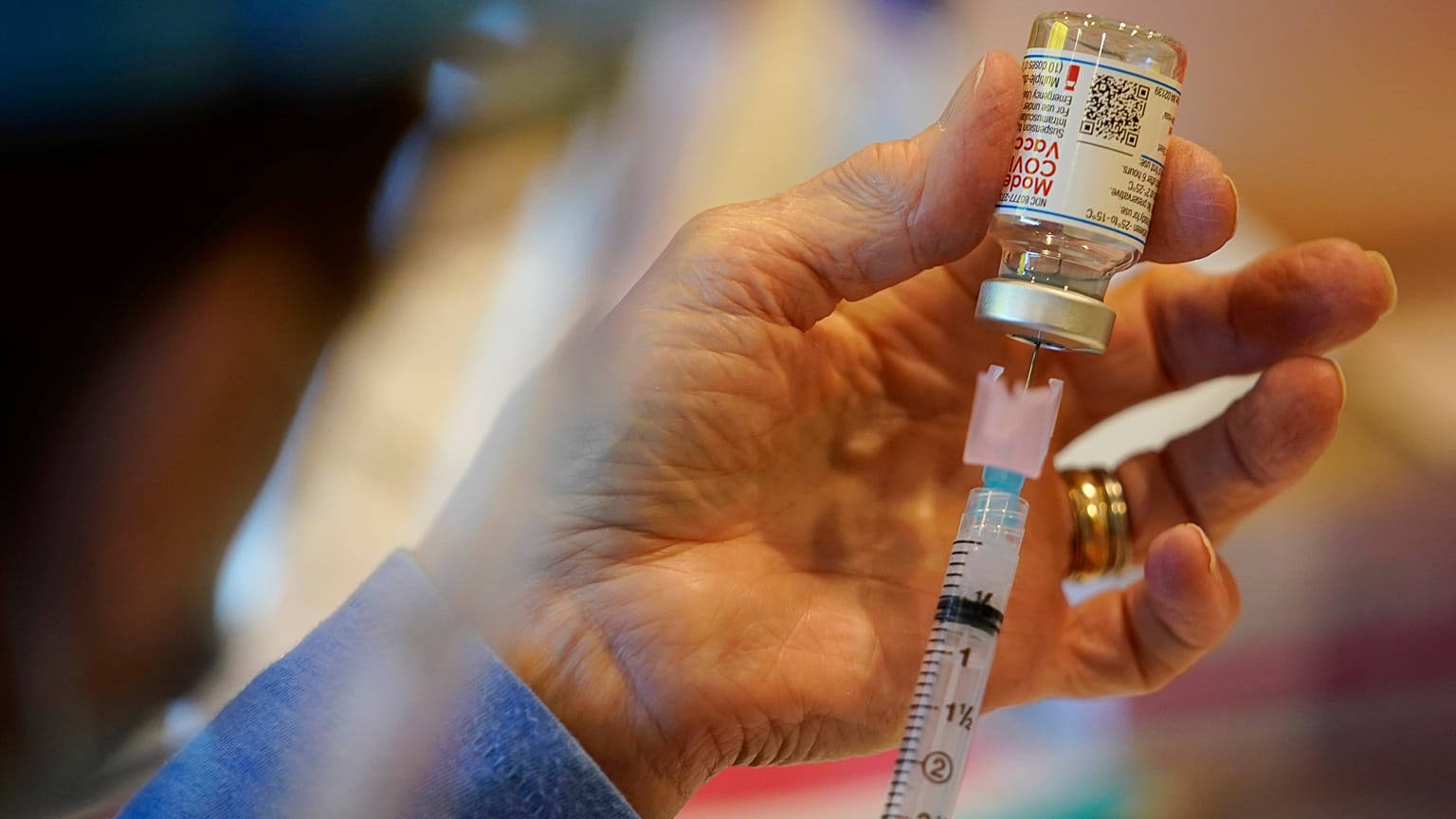
The company plans to submit new data to global regulators.
US biotechnology company Moderna announced on Monday that its COVID-19 vaccine is safe and appears to be effective in children aged 6 to 11.
 ADVERTISEMENT
ADVERTISEMENT
 ADVERTISEMENT
ADVERTISEMENT
The company plans to submit new data from a study of the vaccine in children to global regulators soon.
“We look forward to filing with regulators globally and remain committed to doing our part to help end the COVID-19 pandemic with a vaccine for adults and children of all ages," said Stéphane Bancel, Moderna's CEO.
Moderna tested a lower dose of its vaccine in children aged 6 to 11 to determine it was safe and effective. There were 4,753 study participants who received either the vaccine or a placebo. They received two doses given about a month apart.
The most common side effects were fatigue, headache, fever, and injection site pain.
The company is currently enrolling children 6 months and older for an ongoing study of the vaccine's safety and effectiveness in kids.
The mRNA vaccine was first approved for adults in the European Union in January 2021 and is now available for children aged 12 to 17 as well.
It has not yet been approved for adolescents in the United States.
The COVID-19 vaccine from competitor Pfizer is already being reviewed by European regulators to determine if it will be extended for children between the ages of 5 and 11.
Following mass vaccination with the Moderna and Pfizer jabs, regulators have been monitoring very rare side effects such as heart inflammation, especially in male adolescents and younger adults.