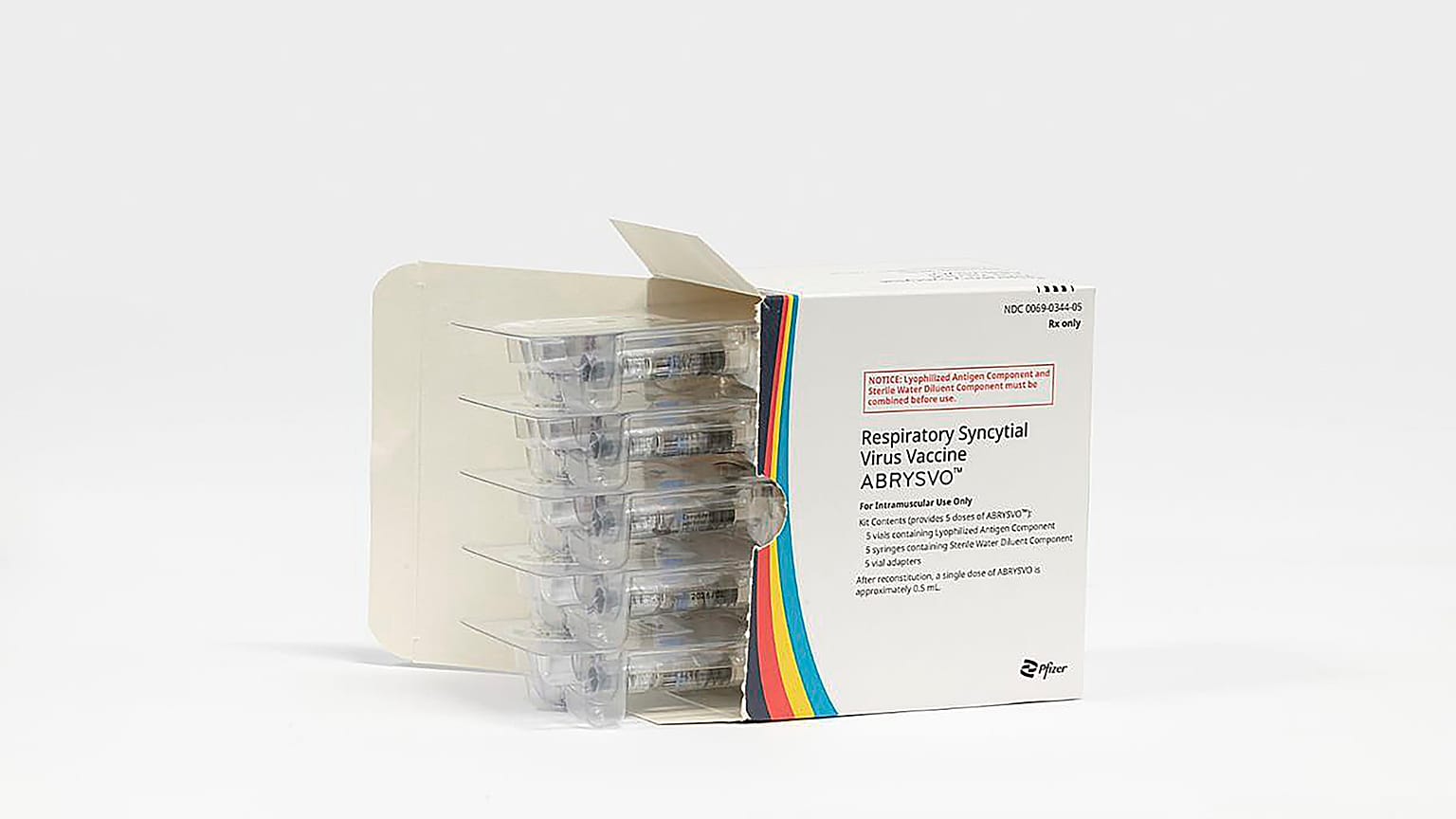
The vaccine was developed by pharmaceutical giant Pfizer.
US authorities approved a vaccine against Respiratory Syncytial Virus, or RSV, for pregnant women for the first time on Monday to prevent a leading cause of infant hospitalisation.
 ADVERTISEMENT
ADVERTISEMENT
 ADVERTISEMENT
ADVERTISEMENT
The vaccine, Abrysvo, has already been recommended by the European Medicines Agency (EMA) for authorisation for infants after their mother is vaccinated during pregnancy.
RSV is a common respiratory illness that causes cold-like symptoms but can be serious for children and adults aged 50 and older.
It can cause bronchiolitis and pneumonia and lead to fatal respiratory distress.
The new authorisation from the US Food and Drug Administration (FDA) is for women who are between 32 and 36 weeks pregnant. Abrysvo is administered as a single-dose injection.
It was previously approved in the US for older individuals 60 years of age and older and has also been recommended for authorisation in this age group in Europe.
Infants among those most vulnerable
"RSV is a common cause of illness in children, and infants are among those at highest risk for severe disease, which can lead to hospitalisation," said Peter Marks, director of the FDA’s Center for Biologics Evaluation and Research.
"This approval provides an option for healthcare providers and pregnant individuals to protect infants from this potentially life-threatening disease".
The most common side effects in pregnant women who received the vaccine in clinical trials were pain at the injection site, headache, muscle pain, and nausea.
The vaccine reduced the risk of severe respiratory illness caused by RSV in infants by 91 per cent when their mothers were vaccinated between 32 and 36 weeks.
In the EU, Norway, and the United Kingdom, RSV causes an average of 213,000 hospitalisations per year of children under five years old, according to the European Centre for Disease Prevention and Control (ECDC).
The EU and US have also recently approved an antibody treatment to prevent RSV infections in infants from pharmaceutical companies AstraZeneca and Sanofi.