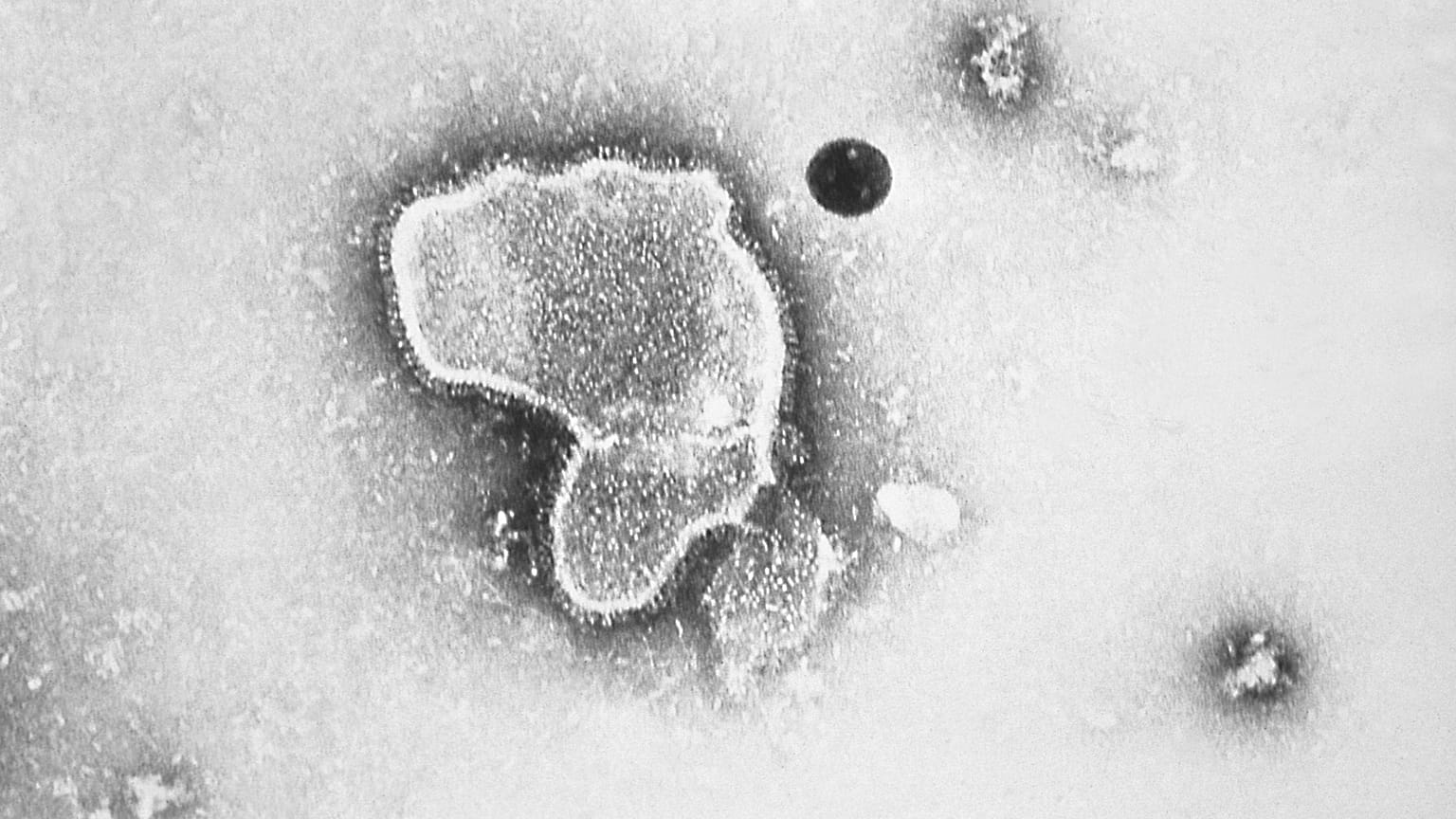
RSV is a common respiratory virus that causes cold-like symptoms but can cause serious illness in children and older adults.
The European Medicines Agency (EMA) recommended authorisation on Friday of a vaccine for infants against Respiratory Syncytial Virus, or RSV, which is a leading cause of paediatric hospitalisation in Europe.
 ADVERTISEMENT
ADVERTISEMENT
 ADVERTISEMENT
ADVERTISEMENT
If authorised by the European Commission, Abrysvo would be the first RSV vaccine that can be administered to infants from birth to six months of age after the mother is vaccinated during pregnancy.
RSV is a common respiratory illness that causes cold-like symptoms but can be serious for children and adults aged 50 and older.
It can cause bronchiolitis and pneumonia and lead to fatal respiratory distress, the EMA said in a statement.
The vaccine authorisation was based on the results of two randomised studies which included thousands of pregnant participants and older adults.
"When a person is given the vaccine, their immune system generates specific antibodies and T cells (immune system cells) that help prevent RSV infection," the EMA said in a press release.
Common side effects for pregnant individuals who received the jab during the study included muscle pain, vaccination site pain and headache. For older adults, vaccination site pain was the single common side effect.
Abrysvo was developed by the US pharmaceutical company Pfizer. The vaccine was approved last month by US regulators for older adults.
Last month, the European Commission approved a different RSV vaccine developed by GSK as its first for older adults.
In the EU, Norway and the United Kingdom, RSV causes an average of 213,000 hospitalisations per year of children under five years old, according to the European Centre for Disease Prevention and Control (ECDC).