Neuralink says it has finally won FDA approval to test its implants in humans, after earlier struggles over safety concerns.
Elon Musk’s start-up Neuralink says the Food and Drug Administration (FDA) has given it the green light to launch its first-in-human clinical study - meaning it will test its brain implant in people’s heads.
 ADVERTISEMENT
ADVERTISEMENT
 ADVERTISEMENT
ADVERTISEMENT
The billionaire’s company says the technology could ultimately help restore people’s mobility and vision by connecting brain signals to computers and allowing a kind of telepathy.
Six months ago, in November, Musk had promised that Neuralink was about six months away from its first human trial, but at the time his pledge had seemed far-fetched as the company was running behind schedule.
Neuralink’s announcement on Thursday, which the FDA did not immediately comment on, would be a critical milestone after the company faced several hurdles on its quest for approval.
The FDA previously rejected Neuralink’s application for human trials over safety concerns, saying there were “dozens of issues” the company needed to address, as reported by Reuters in March.
“We are excited to share that we have received the FDA’s approval to launch our first-in-human clinical study!,” the California-based startup tweeted on Thursday.
“This is the result of incredible work by the Neuralink team in close collaboration with the FDA and represents an important first step that will one day allow our technology to help many people”.
The company added that recruitment for the trial is not yet open, but promised more information “soon”.
How do Neuralink implants work?
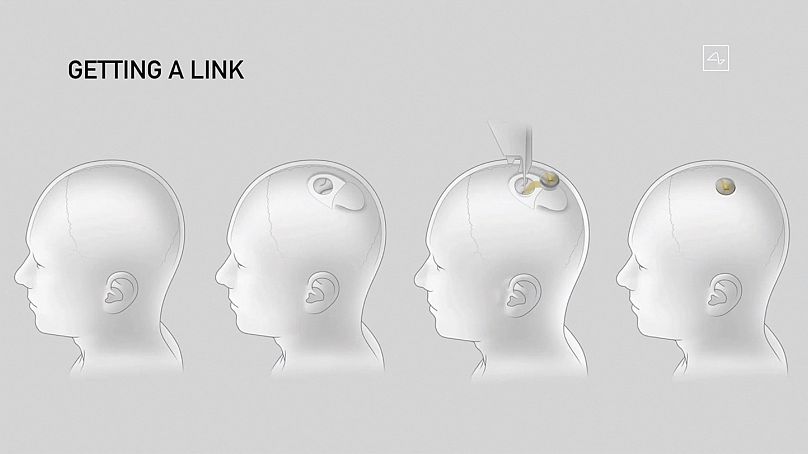
The implant designed by Neuralink - a company Musk co-founded in 2016 -is able to interpret signals from the brain and relay them to devices via Bluetooth, allowing the brain to communicate with computers directly through thought.
The technology - a chip the size of a small coin - has the potential to help people who are paralysed or suffering from neurological diseases to have their sight or mobility restored and be more autonomous.
Last year, Musk said he was so confident in the technology’s safety that he would be ready to implant them in his own children.
Neuralink has already tested its chips in monkeys, a practice which has been the object of controversy over the start-up’s alleged treatment of the animals.
In December, it was reported that the company was under investigation for alleged animal welfare violations, with staff complaining that animal testing was being rushed and needless suffering was being caused to the animals.
The company has denied the claims.
Multiple delays
Through the years, Neuralink has consistently underestimated the time it needed to bring its project to life, initially saying it would start implanting chips in human brains in 2020.
The predicted start of the first-in-human trials was then delayed to 2022 and pushed back again at the end of the year following the investigation into animal welfare violations.
Neuralink’s clinical trial, when it does happen, will not be the first time that a similar brain implant is put inside people’s heads.
In 2021, the FDA greenlighted another company’s request, Synchron, for a clinical trial on humans of its brain-computer interfaces. The first such human implant was completed last July.
Huge progress has been made in this field in Europe too. Thanks to the work of a team of researchers in France and Switzerland, a Dutch paralysed man has regained the ability to walk using only his thoughts thanks to new technology that restored communication between his brain and spinal cord.
No such device has been made commercially available yet.